Nobody Knows What is in the Vials
Covid-19 injections are dangerous, non-compliant biological materials. Their production must be stopped until a full investigation can be done.
Many of us are familiar with the following conundrum: on one hand, highly credentialed scientists and doctors have written numerous research papers explaining the dangerous mechanisms of action underlying mRNA/DNA “platform” technologies. The papers are meticulously researched and depict, correctly in my opinion, many terrifying consequences of the technology that breaches the innate protective mechanisms of human cells. Furthermore, these theoretical papers are validated by the observed outcomes, such as for example, increases in all-cause mortality in high correlation with increases in rates of vaccination in a given territory, unprecedented increases in the adverse events and deaths recorded by various passive reporting (https://openvaers.com/), astonishingly high reports of the adverse events and deaths from the pharmas’ own pharmacovigilance systems, and autopsy findings in vaccinated post-mortem showing the mechanisms of mRNA technology damage in histopathologic evaluations. On the other hand, many who have received the injections report no adverse effects and deem the data points above a “crazy conspiracy”. The question from the uninjured seems to be – why don’t we see MORE deaths if what you say about mRNA products is true? Setting aside ethical limitations of this question, here is a possible answer why:
The mRNA shots do not conform to their label specifications. In practice both “blank” and “lethal” vials and anything in between is produced. Without full compliance, nobody, not even the manufacturers or regulators, know exactly what ends up being produced.
Vials of mRNA injections are not routinely tested by the manufacturers for conformity to the label. The only vial-level tests specified by Pfizer in leaked Chemistry Manufacturing and Controls (CMC) documents are the vial weight at filling, manual inspection for large visible particles, and some tests related to integrity such as vial capping. However, no vial or dose, i.e., “unit-level as dispensed” tests verifying the ingredients are described as routine. How is the public assured that each Pfizer dose contains 30 mcg of mRNA as stated on the label? What level of variability of this key ingredient and other ingredients is acceptable? The ingredient conformity tests described in Pfizer CMC package are based on the bulk product batch testing – an upstream manufacturing process step. It is a regulatory requirement to retain samples of each batch produced, and these samples of vials should exist and be available for examination. Per contracts with the US Government, the product is shipped to the DOD who retains the ownership of the vials until the product is injected into people. These contracts are very detailed and specify manufacturing data to be delivered to the DOD, however, I did not find any descriptions of sampling of the vials for purposes of verification of their contents vs the label. Furthermore, it is expressly forbidden by the international vaccine supply contracts to perform the vial tests for label conformity.
Despite the disturbing prohibition of the independent vial testing, covert random testing of the mRNA vials has been ongoing worldwide. Reported thousands of vials have been obtained and tested by dozens of research groups working independently of each other. The quality of these studies varies and depends on the conditions of the samples acquired, access to the lab equipment and the experience of the investigators. However, the consistent finding among all is that there is yet a single vial to be found in full conformance to the manufacturer’s label. A review of these independent testing efforts has been published recently. Another high-quality report summarizes experiences testing vials from various manufacturers in Germany. These studies use different techniques ranging from optical to electron microscopy, spectroscopy, as well as isolation of genetic and protein components and in some cases sequencing of the RNA.
Some vials contain RNA as well as high concentrations of DNA and protein impurities in quantities far exceeding allowed limits specified by the manufacturer. When RNA was sequenced, the sequences did not fully match the specified BNT162b2 sequence, and a large quantity of RNA fragments was found. In other instances, vials are found apparently without RNA or DNA in them (evidently absent nitrogen and phosphorus).
These results could depend on the methods used and more thorough testing may be needed. Nevertheless, I was able to confirm that the apparent “blank” vials from at least one researcher came from batches of Pfizer and Moderna that had almost no adverse events reports in VAERS: two batch numbers had 1 report each and one batch number had no reports. This should be contrasted with some batch numbers of Pfizer and Moderna associated with 5000+ adverse event reports in VAERS, and an average of ~1500 adverse event reports including ~700 serious reports and deaths across all CDC verified batch numbers.
Almost all vials examined contain high contamination levels of various metals that are toxic to human body. This finding is consistent across all groups and methodologies, and therefore should be deemed more conclusive. There is no explanation of the origin or purpose of these materials according to the known manufacturing processes. Additional findings include various forms of carbon, including, potentially graphene oxide which is a known toxin. Finally, almost all vials examined contain a variety nano- and micro-particulate contaminants – another conclusive finding with plenty of photo and video documentation. These appear under microscope examinations as shapes and structures of various sizes and include characteristic ribbons, fibers, and crystals. Several published reports by qualified and credentialed microscopy experts have excluded the possibility of environmental dirt on the microscope slides. Sometimes a process of movement and what can be described as “self-assembly” is visible and has been documented in a single take video. The researchers also take steps to maintain the chain of custody, examine unexpired product and keep the vials frozen per manufacturer’s instructions.
Below are some representative images from various manufacturers’ injection vials from many studies:
Dr. David Nixon - Australia, Pfizer
Another Dr. from Australia, Pfizer:
The images above are startling and remain unexplained to date. Some scientists insist these are all “salt and cholesterol”, and nothing to see here folks! I disagree. Images of salt and cholesterol under microscope may match some of these structures, but by far do not explain all of them, and seem especially poor explanations for assembly-disassembly processes that have been observed and video recorded. Sizes of these structures alone are problematic. Dismissing them as “manufacturing junk” is not a great way to instill confidence in anyone.
These contaminants and bizarre objects are not rare, in fact as the last image shows, at least some of the vials are teeming with them. While many speculations can be made, one overwhelming conclusion from all the vial tests by independent investigations is that the products are extremely “dirty”, do not conform to their labels and should thus be deemed adulterated. This is a clear indicator that the manufacture of these products is not compliant with the current Good Manufacturing Practices (cGMP).
Sloppy manufacturing process may result from numerous factors, including lack of accountability, negligence, incompetence and possibly fraud. Review of the contracts with the manufacturers made by the U.S. Department of Defense, BARDA and HHS shows that the main driver were the gigantic sums of money made available with no real accountability for quality or safety – all resulting from the forced extreme speed (“warp speed”) of the scale up of manufacturing. The U.S. DOD contracted Pfizer in May of 2020 for production of at least 100 million doses by October 31, 2020, and up to 500 million doses later. Pfizer’s initial contract award was for $10 billion, with many additional incentives for delivering more doses faster. Similar contracts were made with numerous other “vaccine” manufacturers, and hundreds of other suppliers, all under the guise of panic buying for covid countermeasures. No real accountability for product quality or consistency or safety was built into those contracts, in fact the manufacturers were explicitly exempt from all possible liability under the PREP Act, which is specified in a separate contract clause. The purchaser is the DOD, the distributor is the DOD (and not the licensed and accountable pharmaceutical distributors), and furthermore, the product is not serialized and hence open to both adulteration and falsification or mislabeling.
To meet the contract obligations, Pfizer’s manufacturing batch size has increased from microgram scale for lab and animal study volumes to commercial batches of ~140 liters at the end of 2020 and ~300 liters by late 2021. In my experience commercializing biotechnologies from academia, failure to scale is one of the leading causes of failures of all new technology ventures. Showering this problem with money rarely accelerates the solution, and very often accelerates the demise of the whole venture. This is common sense. For example, placing an order today for 1,000,000 of a new type of vehicles with Ford Motor Company to be delivered in 6 months will likely fail no matter the dollars spent, since even obtaining the raw materials in time will be problematic. Complex manufacturing requires materials, systems, capacity, experienced staff, established processes, suppliers, and most importantly control systems at the correct scale to be successful and produce high quality consistent product. Now imagine simultaneously asking several direct competitors - Ford, GM and Toyota to produce 1,000,000 new cars each in the next 6 months.
Based on review of available literature on mRNA manufacturing and my discussions with experts who have made mRNA in the lab, it is not clear that mRNA can be manufactured at the scale that is estimated for these injections from known shipment numbers and disclosed manufacturing documentation: 200-300 liters of drug product per average batch, 700+ batches a year in the US alone. This is particularly unlikely if strict cGMP rules are applied to the manufacturing requirements, and we know that cGMP is not followed for production of these injections. Recently FDA found Catalent non-cGMP compliant. Catalent handles large volumes of fill-finish for Moderna, therefore batches processed through Catalent are non-cGMP compliant.
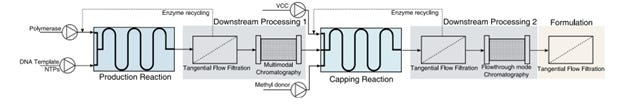
The generation of mRNA by in-vitro transcription (IVT) at large scale and under current good manufacturing practice conditions is challenging. For example, the specialized components of the in-vitro transcription IVT reaction must be acquired from certified suppliers that guarantee that all the material is animal component-free and GMP-grade. Furthermore, the availability of large amounts of these materials is limited and purchasing costs are high. This is true, for example, in the case of the enzymes used for translation and capping. Even the glass vials themselves were reported as a shortage. Additionally, the low yields and batch failure are a known problem. Conceptual process flow of making mRNA drug substance contains several steps:
The process is composed of a 2-step enzymatic reaction in continuous form, followed by enzyme recycling using tangential flow filtration strategies and two multimodal chromatography steps, one in bind-elute mode for the intermediate purification, and a second in flowthrough mode for polishing. Formulation is achieved using a third tangential flow filtration module. This means the mRNA needs to be made by chemical reaction, and then purified, and then capped and purified again. There are many variations of this process, and no standards exist. At the “formulation” step (last box in the picture), there are further multiple steps to create lipid nanoparticles, and get the mRNA encapsulated. Further, there are fill and finish steps that likewise are not problem free and decrease the yield. Finally, transportation and manual dose preparation add an extra variability layer.
Here is a simple heuristic to understand any manufacturing process flow and not get confused by the jargon: each arrow in the flowchart points to places where errors accumulate Each output-input point in a complex manufacturing flow is where the errors can be checked for and rectified or, if unsolved, will amplify and destroy the product quality and consistency. This is especially dangerous at the extreme speed and scale.
In science papers mRNA manufacturing is described with problem-free cartoons, it all works beautifully regardless of whether it is microgram or kilogram output, and not a single paper on this topic dwells much on low yields or process failures. This is because the academia never has to confront reality. However, the pesky reality of manufacturing mRNA (or anything else) at large scale is highly error prone. Each step has a yield of anywhere between 50% and 80%, and sometimes the whole batch fails, and that is especially true at the large scale of production. mRNA reaction fidelity is less than 100%, the caps and tails fall off, mRNA breaks into fragments, lipid nanoparticles do not form perfectly and PEGylation can be inhomogeneous leading to their breakage and subsequent escape and breakage of mRNA. Large mRNA breaks into smaller fragments, and these remain in the substance. At large scale of reaction, the enzymatic process for making mRNA was reported seizing at 37.5 liters of mRNA substance (before encapsulation into LNP and making drug product) according to the European Medicines Agency (EMA) documents. It is not clear how this was resolved and transitioned to 100's of liters scale in a matter of few months, and for all suppliers. mRNA fragments were deemed process related impurities by the EMA who raised a significant concern, but they were dismissed by the FDA as a “theoretical problem” - as leaked emails from EMA have shown. mRNA “fragments” may or may not code for proteins, however micro-RNAs (miRNA) can interfere with endogenous cellular processes to detrimental health effects which is described in many scientific papers and even in a textbook on biological weapons published by the NIH in 2018. Chapter 6 of the book describes gene therapy as a class of biological weapons. Coincidentally, the DOD-pharma contracts for covid-19 shots also explicitly state that the product is being developed for both civilian and military applications.
Returning to mRNA manufacturing, multiplying even an optimistic 80% yield by, for example, 7 process steps results in 20% final yield, and if the in-process failures are larger, final yield is single digit percentage or a failure has occurred. Each step generates large amounts of impurities, which are never fully removed as aggressive purification will break the fragile product. Furthermore, the mRNA substance is never equally distributed in the batch volume as thorough mixing of the product is not possible due to its fragility, and lipids tend to float to the top of the vat as well as stick and congregate together. Dangerous possibly cytotoxic aggregates of broken LNPs and mRNA (mRNA adducts) can result and were reported by Moderna a full year after large scale deployment of their product. As a result, the larger the volume of the batch, the more inhomogeneity at the vial level. These conditions can create over-concentrated, toxic vials and the ones that could be “blanks”, or anything in between. The larger the batch volume the more duds it will produce, which in case of this product is largely good news for the injectees. Avoidance of specifying any product conformity tests at the vial level by the manufacturers seems to be intentional in this context.
Data from Pfizer’s own CMC documentation submitted to EMA at the end of November 2020 shows “failure to scale” beginning at approximately 25% of the current commercial scale of the batch (current scale = 600,000 vials = 3-4 million doses per batch). The graphs below were generated using exact sizes in vials and doses for each Pfizer batch manufactured between August and November 2020 (Figure 1) and all Pfizer shipped doses in the US up to end of April 2022 (Figure 2). I used reported deaths and adverse events in VAERS database associated with those batches as a measure of batch variability. This does not address vial variability but provides directional information especially for the scale of manufacturing. The batches in Figure 1 were the first ones to ship commercially and were likely all used close to 100% since at that time the demand for these injections was insatiable. As the “scale-up” of manufacturing proceeded in 2020, the batches were manufactured in a variety of sizes from 50,000 and up to 300,000 vials (~140 liters of drug product). During this time, several major changes were made to the manufacturing, for example, transition from the pilot facility at Polymun Scientific to Pfizer’s own plants and changing major manufacturing steps to new processes.
Figure 1.
First, the overall data indicate a statistical trend toward increased number of reported deaths with the increase in the batch size – the more this product is used, the more deaths are reported. This is not news for anyone who has been paying attention to the injection related adverse events and deaths. However, the variability batch-to-batch demonstrated by the vertical dispersion of the death reports associated with batches of the same size is already apparent at approximately 150,000 vial batch size (25% of the full commercial scale). Batches of the same size are 4-5 times different from each other in the number of reported deaths.
The “failure to scale” story gets larger at scale, no pun. Recently, the exact sizes of Pfizer lots shipped in the United States became known via a FOIA data release, including all doses with associated lot numbers shipped as of end of April 2022 to various vaccination centers. Figure 2 is the plot of all batches from Pfizer, by their reported size in doses on the x-axis and serious adverse events including deaths reported for those lot numbers on the y-axis. Data from VAERS was downloaded on September 24, 2022.
Figure 2.
This graph includes the “early” scale up batches from Figure 1, as well as what appears smaller shipments possibly for batches that were largely distributed overseas. What is immediately apparent from the data – the staggering inconsistency of the product batch-to-batch and the rapid decline of apparent toxicity measured by the adverse events with increase in batch size. The latter trend is the opposite of what was observed with the early batches. The product is causing fewer adverse events per dose when there are more doses available. This does not make sense, except if these doses are simply sitting on the shelves. Of note is batch FM0173 (only 26,700 doses shipped in the US) that generated the highest rate of serious adverse events (3.3/1000 doses), upper left dot.
The scale of manufacturing strains the credulity. A batch of 12 million doses translates to approximately 900+ liters of mRNA! Given the manufacturing steps involved, impurities generates and the amount of raw materials, and the scale of manufacturing equipment and disposables needed, it should be questioned whether this truly was a single production run.
Figure 3 is the same data as in Figure 2, with outlier batches removed for clarity (includes batch sizes from 100K to 4M doses):
Figure 3:
It is evident that the variability batch-to-batch is highly significant and remains unexplained. It is also strangely declining as the size of the batch is becoming larger. The only reasonable explanation to this is that the usage of this product per batch has plummeted. Most of what is produced later in the time period is sitting on the shelves. At the end of the time period in this graph (end of April 2022), an estimated 100M doses were manufactured but not administered in the United States. At the beginning of this graph, nearly all manufactured doses were administered. That is the only reasonable explanation – and it proves that these injections cause the injuries and deaths reported to VAERS!
Table below lists all batches >4 million doses, including their date of manufacture and the number of serious adverse events and deaths reported for them in VAERS:
Table 1:
The “mega” batches are not entirely benign. They are simply a larger lottery pool. The single death reported for batch FL0007 is for an 8-year-old girl who died in Texas from a multi-system organ failure (VAERS ID 2327226-1) – see Figure 4. While the first batch listed in her report RL0007 appears to be a typo (RL series do not exist for Pfizer), it is evident that she received both doses from the same FL0007 batch and passed away 3 months later.
Figure 4:
Here is my educated guess on what is going on with the batch variability: Pfizer’s (and other manufacturers’) contracts were for delivery of DOSES. Millions of them and fast. Contract scope is simply a “demonstration of large-scale manufacturing” and billion-dollar bonuses attached for shipping millions of doses by certain dates. No accountability, no checks, no liability, just ship the doses on time! The batches of 5 million+ doses should be questioned in this context. These appear largely benign from the adverse event perspective but, since the demand for these shots has plummeted coinciding with production of mega-batches, it is hard to say what the real driver of “safety” is – over-dilution of the product or refusal of the customers to be injected. I hope it is the latter.
Here is as close as I can get to answering the question “why aren't MORE people dying?”: Too many people have died and have been injured by these injections, and plenty more will ultimately have their lives cut short. The manufacturers are making both - lethal shots and highly diluted “blanks” in a sloppy, uncontrolled, unaccountable, and ultimately fraudulent manner.
To know the truth, these products must be tested at the vial and dose levels, in a random sampling by independent 3rd party laboratories. In the meantime, the products must be recalled, and a proper investigation initiated.
Art piece for today: The Baker, oil on panel, 24x18 inches. The scene for this painting is from a pastry shop in Ukraine.
















Just finished the article. This was phenomenal work, Sasha. Immense gratitude to you for writing it.
God bless!
Thank you Sasha for your doggedness, diligence and the clarity that you provide.